Kwaliteit is altijd al een cruciale factor geweest in de productie, maar vandaag de dag is kwaliteitsmanagement belangrijker dan ooit, omdat klanten meer keuzemogelijkheden hebben en gemakkelijker toegang tot informatie over concurrerende producten. Fabrikanten hebben toegang tot hulpmiddelen om kwaliteit en efficiëntie in hun processen in te bouwen, bijvoorbeeld software oplossingen zoals manufacturing execution systems (MES), warehouse management systems (WMS) en quality management systems (QMS). Hoewel elk van deze systemen zorgt voor zichtbaarheid, tracking en inzichten die leiden tot meer kwaliteitscontrole en beheer, kan de implementatie van verschillende uiteenlopende tools leiden tot vertragingen en problemen bij de productie, waardoor het doel van de individuele oplossingen teniet wordt gedaan.
Hoe een volledig geïntegreerde oplossing OEE kan verbeteren
Zonder geïntegreerde systemen voor supply chain- en kwaliteitsbeheer krijgen fabrikanten te maken met mogelijke vertragingen in de productie doordat zij handmatig gegevens moeten invoeren of een apart volgsysteem moeten onderhouden, zoals het verzamelen van gegevens op papier of spreadsheets. Deze vertragingen verspreiden zich, waardoor de doorlooptijd van inspecties en QC-bemonstering toeneemt. Het loskoppelen van productie en opslag en logistiek verergert de kwaliteitsproblemen nog verder, wat leidt tot verspilling en verdere vertragingen.
Gebrek aan integratie leidt tot een domino-effect, met als gevolg dat er meer voorraad aanwezig moet zijn om tekortkomingen in de kwaliteitscontrole en extra kosten en verspilling van waardevolle opslag- en productmiddelen te compenseren.
Maar belangrijker is dat afzonderlijke systemen werken tegen de algemene efficiëntie van de organisatie. Afzonderlijke software-oplossingen leiden tot een silo-afdeling en een “wij tegen hen” mentaliteit tussen productie, kwaliteitscontrole en logistiek. Hoewel deze situatie niet optimaal is voor de efficiëntie van de organisatie, resulteert het ook in QC-goedkeuring van niet-goedgekeurde producten, handmatige inspanningen bij het testen van rapporten die inefficiënt en minder nauwkeurig zijn en die de overgang naar papierloze productie belemmeren – een primair doel van MES-softwareoplossingen. Papierloze productie centraliseert alle informatie die verband houdt met de productie en ondersteunt continue verbetering en slanke productie-initiatieven.
Problemen met afzonderlijke systemen
Zonder geïntegreerde systemen voor supply chain- en kwaliteitsbeheer krijgen fabrikanten te maken met mogelijke vertragingen in de productie doordat zij handmatig gegevens moeten invoeren of een apart volgsysteem moeten onderhouden, zoals het verzamelen van gegevens op papier of spreadsheets. Deze vertragingen verspreiden zich, waardoor de doorlooptijd van inspecties en QC-bemonstering toeneemt. Het loskoppelen van productie en opslag en logistiek verergert de kwaliteitsproblemen nog verder, wat leidt tot verspilling en verdere vertragingen.
Gebrek aan integratie leidt tot een domino-effect, met als gevolg dat er meer voorraad aanwezig moet zijn om tekortkomingen in de kwaliteitscontrole en extra kosten en verspilling van waardevolle opslag- en productmiddelen te compenseren.
Maar belangrijker is dat afzonderlijke systemen werken tegen de algemene efficiëntie van de organisatie. Afzonderlijke software-oplossingen leiden tot een silo-afdeling en een “wij tegen hen” mentaliteit tussen productie, kwaliteitscontrole en logistiek. Hoewel deze situatie niet optimaal is voor de efficiëntie van de organisatie, resulteert het ook in QC-goedkeuring van niet-goedgekeurde producten, handmatige inspanningen bij het testen van rapporten die inefficiënt en minder nauwkeurig zijn en die de overgang naar papierloze productie belemmeren – een primair doel van MES-softwareoplossingen. Papierloze productie centraliseert alle informatie die verband houdt met de productie en ondersteunt continue verbetering en slanke productie-initiatieven.
Een closed-loop oplossing om QM-processen te integreren
In plaats van te vertrouwen op een handvol afzonderlijke oplossingen – en zich zorgen te maken over de risico’s van uiteenlopende systemen – zouden fabrikanten moeten overstappen op een gesloten, volledig geïntegreerde oplossing voor het beheer en het volgen van de overall equipment effeciency (OEE) en de kwaliteit van gate tot gate. Door integratie kunnen fabrikanten hun productiviteit verhogen en een hoger kwaliteitsniveau garanderen. Integratie elimineert de problemen die gepaard gaan met afzonderlijke systemen, zoals onnauwkeurige of onvolledige registratie en een gebrek aan zichtbaarheid van de ene afdeling naar de andere.
Een closed-loop systeem bevat informatie voor een betere nauwkeurigheid en zichtbaarheid voor een holistische aanpak. Met de mogelijkheid om nauwkeurige, real-time informatie te zien in elk stadium van de productie of kwaliteitscontrole, hebben alle correctieve en preventieve acties meer betekenis, omdat een consistente toepassing van deze acties kan worden gegarandeerd.
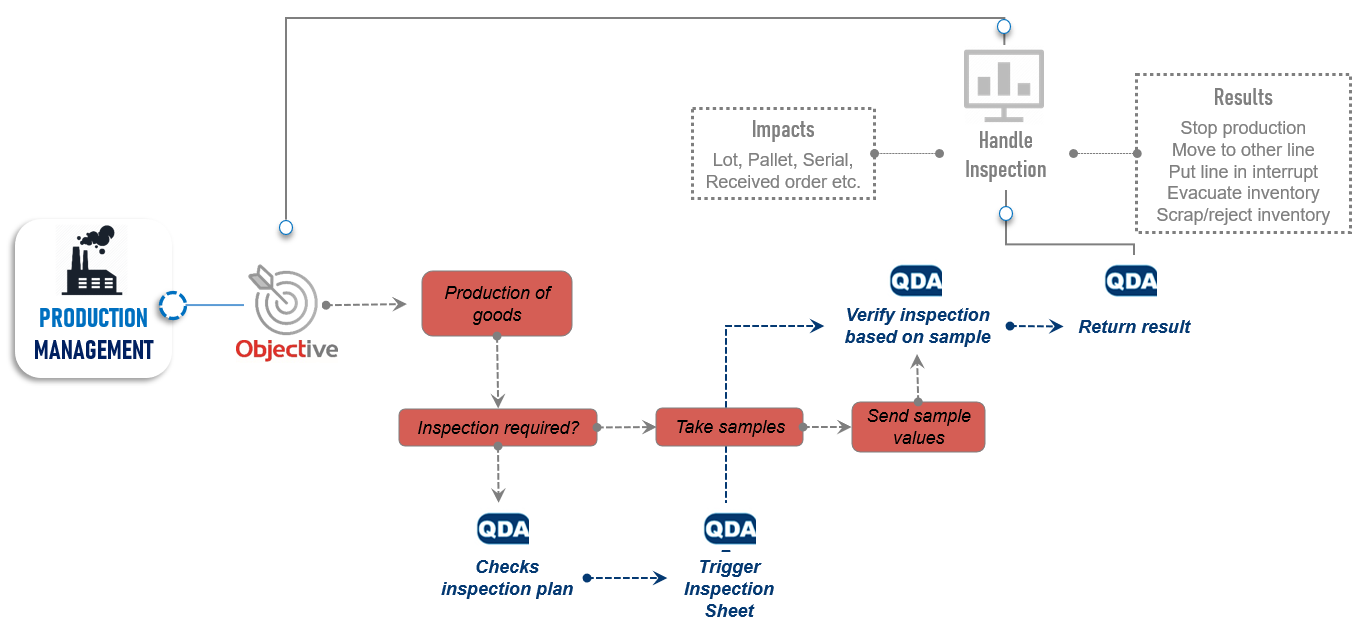
Consistente prestaties neerzetten
De MES-oplossing van Objective integreert met QDA QMS en helpt fabrikanten een consistente en nauwkeurige controle van hun gehele productie, kwaliteitscontrole en logistieke processen te behouden. Het geïntegreerde systeem omvat OEE, logistiek/magazijn, gegevensverzameling, laboratoriuminformathttp://www.objt.com/en/wms-voor-de-maakindustrie-discrete-manufacturingiesystemen (LIMS), non-conformiteitsmanagement, geïntegreerde kwaliteitsplanning, supply chain management en nog veel meer. Van verminderde voorraadvereisten tot minder logistieke bewegingen, fabrikanten ervaren ook kostenbesparingen.
De oplossing helpt niet alleen bij de efficiëntie, maar vereenvoudigt ook de audits en is eenvoudig te integreren met andere systemen, zoals ERP-oplossingen en PLC/SCADA-machines. Door deze integratie kunnen fabrikanten de kosten verlagen en de productkwaliteit verbeteren.
De consistentie van het closed-loop systeem zorgt ook voor een enkele gestandaardiseerde configuratie voor het gebruik op meerdere locaties en voor 24/7 systeemondersteuning. Met Objective en QDA SOLUTIONS kunt u alle kwaliteitsbehoeften van het ontwerp en de ontwikkeling tot aan de levering en distributie behandelen en een globaal beeld van de kwaliteit van uw bedrijf in één oplossing bereiken.
Het resultaat is een naadloze, nauwkeurige en volledig transparante productie, zodat fabrikanten de status van hun processen kennen en nu en in de toekomst nauwkeuriger en effectiever resultaten kunnen leveren. Fabrikanten kunnen hun doelstellingen op het gebied van operationele efficiëntie en kwaliteitsmanagement halen, omdat de expertise op alle afdelingen, van productie tot magazijn en kwaliteitscontrole, volledig kan worden benut.
Voor meer informatie over hoe de geïntegreerde kwaliteitsmanagementsystemen van de oplossing de efficiëntie kunnen verhogen, de prestaties kunnen verbeteren, het concurrentievermogen op de markt kunnen handhaven en de klantervaring kunnen verbeteren, kunt u contact met ons opnemen voor meer informatie.



